Executive Summary
The gap in visual inspection for difficult-to-inspect injectable products is widening as biologics and lyophilized injectables reshape pharmaceutical manufacturing. Contract manufacturing organizations (CMOs) are being asked to inspect inherently variable products (powders, cloudy or textured cakes; molded glass, vials, syringes) using systems originally designed for clear liquids. The regulatory landscape is also evolving; 75% of recent FDA complete response letters (CRLs) were tied to quality and manufacturing deficiencies. A single CRL can delay approval by years, deferring billions in revenue and placing intense pressure on manufacturing partners. Meanwhile, traditional inspection options carry steep tradeoffs: manual and semi-automated inspection are hampered by fatigue and inconsistency, and traditional automated systems are inflexible and difficult to qualify for complex products. For CMOs operating on thin margins, inspection of variable products has become a direct financial, operational, and reputational risk. This white paper covers the structural drivers behind this inspection gap and defines the evaluation criteria CMOs should use to mitigate regulatory risk and protect margin.
The Visual Inspection Challenge Faced by CMOs
The pharmaceutical industry is in the midst of a structural shift toward biologics. Monoclonal antibodies, mRNA vaccines, and targeted oncology therapies are among the fastest-growing drug classes in development. Unlike small molecule drugs, these newer drugs are extraordinarily fragile in liquid form. By removing water and converting the product into a freeze-dried cake, a process known as lyophilization, manufacturers can extend shelf life and get therapies to market faster. But manufacturing a lyophilized injectable poses unique challenges. Unlike other parenterals, the lyophilization process results in a powdery substance that can greatly vary in texture, color, and intensity. Traditional visual inspection systems that were built for clear liquid products are unable to detect glass, metal, and other foreign particulates in these products with any degree of confidence, turning inspection of advanced pharmaceuticals into a direct regulatory risk.
That risk is being felt across the industry. Contract manufacturers are operating in a fundamentally different environment than they were just five years ago. Recent data published by the FDA reveals that 75% of complete response letters, the agency’s mechanism for blocking a drug from reaching the market, are tied to quality and manufacturing deficiencies. According to RSM, a CRL delays a drug’s final approval by 2.5 years on average, causing billions in deferred revenue. As a result, biopharmaceutical companies are no longer sourcing manufacturing capacity primarily on cost. Instead, they are shopping for regulatory certainty, and they expect their CMO partners to deliver it.
CMOs are caught between a rock and a hard place. Manufacturing sponsors want tighter quality-assurance processes to prevent CRLs. But the products CMOs are being asked to manufacture are too difficult for traditional visual inspection techniques.
Current State of Visual Inspection in Contract Manufacturing
Within contract manufacturing organizations, end-of-line visual inspection has traditionally been carried out in one of three ways: manual, semi-automated, or fully automated.
Manual Visual Inspection
Manual visual inspection remains the gold standard. Inspectors evaluate one unit at a time against alternating black and white backgrounds. It’s flexible, requires a modest investment in equipment, and is favored by regulators. The process, however, is slow, labor-intensive, and subject to the one variable no training program can fully control: human fatigue. Performance degrades over a shift, accuracy varies from inspector to inspector, and scaling output means scaling headcount.

Semi-Automated Visual Inspection
Semi-automated visual inspection (SAVI) increases throughput without fully removing the human from the process. Units pass through a machine at 10 to 30 per minute, with optimal lighting and a pre-spin to mobilize particulates before an operator makes the final accept or reject decision.
CMOs often prefer to use SAVI for lyophilized products and other difficult-to-inspect parenterals because it combines automated material handling with the adaptability of human inspectors. However, the added speed comes at a cost. With only seconds to evaluate each passing vial, semi-automated visual inspection can be highly inconsistent and prone to error.

Automated Visual Inspection
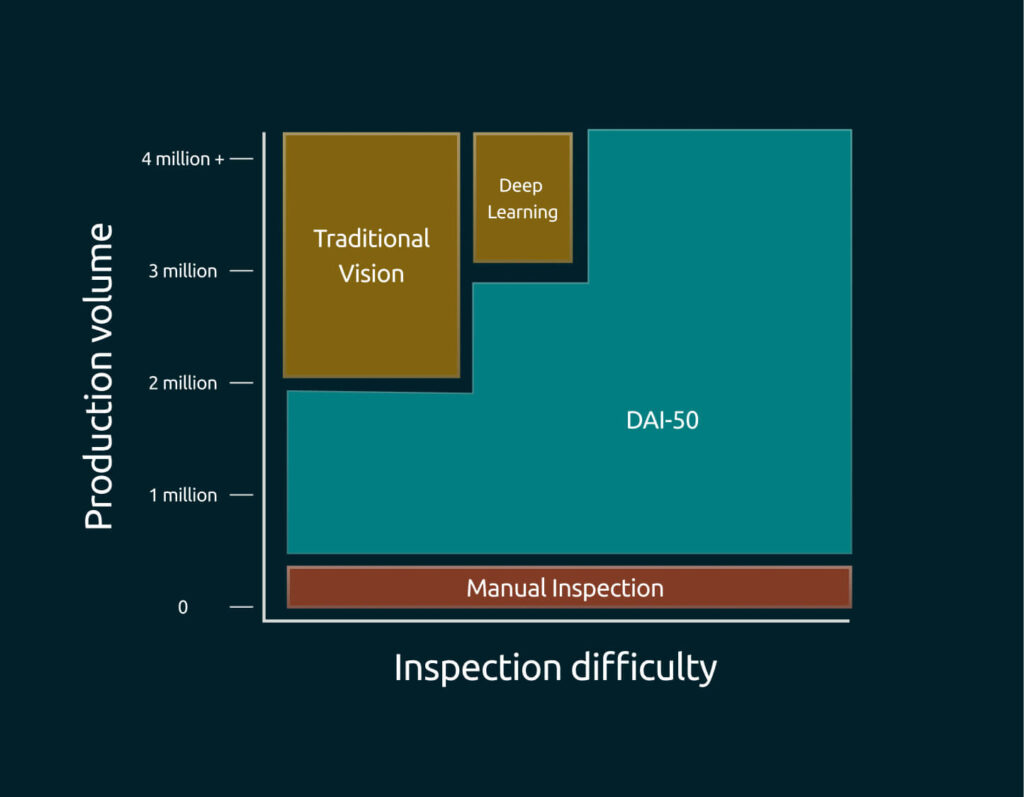
Traditional automated visual inspection (AVI) systems use a combination of automated material handling, computer vision, and rule-based defect detection algorithms (step-by-step if/then statements) to inspect up to 600 units per minute. AVIs are typically used for clear liquid products manufactured in large batches exceeding 10 million units. However, when applied to difficult-to-inspect products, traditional AVIs can have a false reject rate of up to 30%. In addition, their robust mechanical configuration makes it time-consuming to change between different product SKUs and nearly impossible to switch between a vial and a syringe.

AI-Based Visual Inspection
In response to the limitations of traditional rule-based systems, visual inspection original equipment manufacturers (OEMs) have invested millions in AI-based visual inspection to close the accuracy gap. Most of these solutions rely on supervised machine learning, where neural networks are trained on thousands of labeled defect images to learn specific, predefined defect categories. A supervised deep learning model can improve detection performance for known defects, helping to reduce some of the rigidity associated with traditional rule-based algorithms.
However, this approach comes at a cost. True defects in pharmaceutical manufacturing are rare by nature, making it extremely difficult and impractical to collect the thousands of consistent, labeled defect images required to train a model. Even when sufficient data is gathered, models must be retrained for each container type, product variation, or new defect category. Based on our research, only a limited number of inspection systems that rely primarily on supervised machine learning have been successfully deployed and qualified in production.
Why the Legacy Approach to Pharma Visual Inspection Isn’t Sufficient
The Gap in Visual Inspection Equipment
There is a growing gap in pharmaceutical visual inspection between what traditional systems promise and what modern manufacturing actually requires. As product portfolios become more complex, conventional rule-based vision systems and supervised machine learning approaches struggle to keep pace. Supervised models require thousands of labeled defect images per category, an unrealistic expectation in an environment where anomalies are rare and often novel . Even when defect data can be generated or synthetically created, models frequently prove brittle and fail when exposed to real-world variability. Meanwhile, manual inspection remains inconsistent, fatigue-prone, and difficult to scale.
The result is an industry caught between slow, unreliable human inspection and expensive, inflexible automation that cannot adapt to unknown defects. For CMOs, this gap translates directly into financial and operational risk:
- Elevated false reject rates that erode yield.
- Acceptable quality limit (AQL) failures that delay batch release.
- Reputational exposure with sponsors and regulators.
- Rising labor costs driven by the need to continually scale and retrain inspection staff.
Financial Impact for Contract Manufacturing Organizations
Contract manufacturers operate on razor-thin margins with zero room for waste. Unlike branded pharma companies that absorb inspection costs across massive production volumes, CMOs bear the financial burden of every rejected vial, every failed AQL, and every hour spent reinspecting a product that should have passed the first time. The compounding effect of these inefficiencies can erode profitability and put client relationships in jeopardy.
High False Ejects
Every unit falsely rejected represents missed revenue and added production costs. This burden is particularly acute for contract manufacturing organizations. A branded manufacturer producing a single high-volume product can refine one process and distribute inefficiencies across millions of units. A CMO managing dozens of SKUs for multiple clients operates under far greater complexity. Every product has its own inspection parameters, its own false reject profile, and its own margin implications. As a result, elevated false reject rates compound operational strain and erode profitability across the portfolio.
Failed AQLs and Reinspection Costs
A failed AQL can trigger a number of costly downstream activities: root cause investigation, reinspection of the entire lot, additional documentation, and in some cases a complete scrap of the entire lot. For CMOs handling sterile injectables with limited shelf life, these delays compress already tight delivery windows and can result in contractual penalties. Every reinspection cycle pulls qualified personnel and equipment away from scheduled production, creating bottlenecks that ripple across the facility.
Brand Management and Reputation
Trust is at the center of the relationship between the CMO and its sponsors. A single quality event can permanently alter a client’s perception of a facility’s capability. In an industry where switching costs are high and qualification timelines are long, most clients will tolerate occasional issues. But a pattern of inspection-related malperformance raises serious questions about process control, technology investment, and operational maturity. A CMO’s audit performance and quality metrics are often the deciding factor in competition for new business, and chronic inspection problems leave a paper trail that’s difficult to explain away during a client audit or regulatory review.
Rising Cost of Manual Inspection
Manual and semi-automated inspection have long been the default for difficult-to-inspect products, but the economics are shifting. Labor costs continue to climb, trained inspectors are difficult to recruit, and the time required to qualify new personnel can be cumbersome. A team of human inspectors can easily represent hundreds of thousands of dollars in annual labor. For CMOs experiencing price pressure from clients and margin compression from rising material costs, the manual inspection model is becoming harder to sustain with every passing quarter.
What to Look For in a Pharmaceutical Visual Inspection System
For contract manufacturing organizations evaluating new visual inspection technologies, the decision goes beyond throughput alone. Inspection systems directly impact product quality, regulatory compliance, operational efficiency, and overall profitability. To make an informed decision, CMOs should assess visual inspection solutions across three key criteria: flexibility, accuracy, and total cost of ownership.
Flexibility and Product Changeover
In high-mix environments, time lost to product changeovers directly impacts revenue. Leaders should evaluate how quickly and predictably a system can transition between SKUs without prolonged downtime or reliance on OEM support. For CMOs, agility matters as much as throughput.
Key questions to ask:
- How long does it take to switch between product sizes or container formats?
- How complex are the mechanical adjustments required during changeover?
- Does the system rely on OEM technicians or specialized engineers for routine changeovers or ongoing operation?
- What changes parts are required other than parts, and what do they cost?
- Can the system inspect different types of primary containers (vials, syringes, bottles, etc.)?
- Can the system inspect different types of product formulations (powders, lyophilized products, opaque liquids, etc.)?
- Does adding a new SKU require new hardware, extensive programming, or retraining?
Accuracy and False Reject Rates
Visual inspection systems must balance defect detection with yield protection. High false reject rates erode margin, increase reinspection costs, and create operational bottlenecks. Executives should assess real-world false eject performance, stability over time, and comparative results versus manual inspection benchmarks. Ultimately, a high-performing system must strike the right balance between sensitivity and specificity to ensure product quality without unnecessarily sacrificing yield.
Key questions to ask:
- What is the system’s probability of detection across different defect categories?
- What false reject rate does the system achieve under real production conditions—not just controlled testing?
- How consistent and repeatable is performance across shifts, batches, and extended runs?
How does the system’s performance compare to manual inspection benchmarks?
Total Cost of Ownership
Capital expense is only the starting point. True cost includes labor requirements, retraining, maintenance, model tuning, validation cycles, and scalability as demand fluctuates. Executives should evaluate the total cost of ownership to understand the system’s impact on margin over time.
Key questions to ask:
- What are the ongoing labor requirements associated with operating and supporting the system over time?
- What level of maintenance and service does the system require to sustain performance?
- How frequently must the system be revalidated or requalified?
- How do costs scale as production demand increases or decreases?
Conclusion
The pharmaceutical industry has outgrown legacy visual inspection. Manual inspection has poor economies of scale. Semi-automated systems lack accuracy. Traditional rule-based systems are too expensive and produce unacceptably high false eject rates. And solutions based on supervised machine learning depend on defect libraries that are impractical to build and incapable of detecting novel defects. As product portfolios continue to shift toward difficult-to-inspect formats, CMOs seem to be left with a choice between inconsistent human inspection and inflexible automation. Advances in unsupervised machine learning, however, can now help address these challenges.
The DAI-50, powered by AVIS, closes the gap by delivering automated visual inspection with the reliability of a machine and the flexibility and accuracy of the best human inspector. Using unsupervised machine learning, AVIS models normal product variation directly from 500 pre-inspected compliant units, eliminating the need for labeled defect libraries and enabling detection of both known and previously unseen anomalies. The result is a scalable inspection process that increases throughput, protects yield, and delivers the regulatory confidence sponsors expect from a contract manufacturing partner.


