AI-Based Visual Inspection Machine for Injectable Products
Enhance product quality and 3x visual inspection speeds with our AI-based visual inspection machine for injectable products.
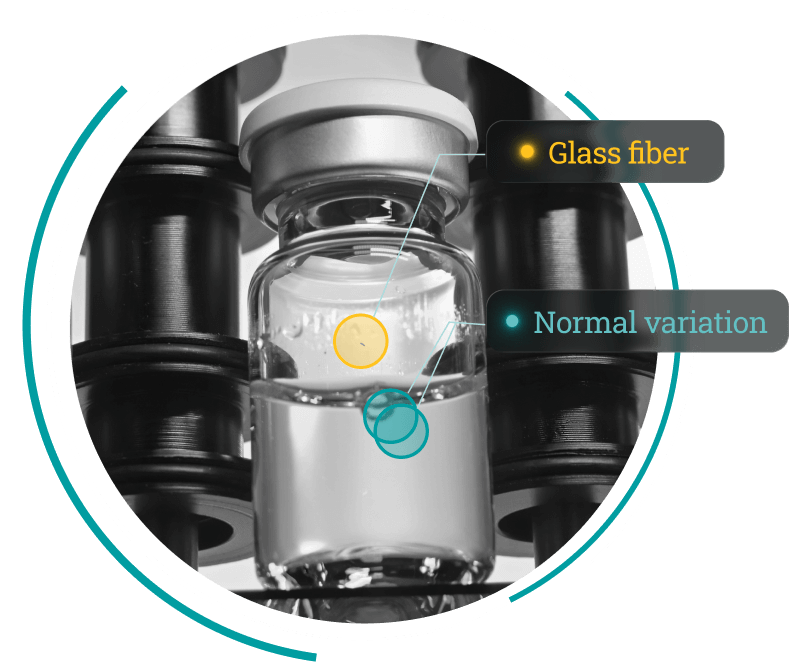

Injectable Products Supported by our AVIS Platform
AVIS is the first visual inspection system designed for difficult-to-inspect injectable products. Regardless of your visual inspection challenge, AVIS can accurately detect anomalies and reduce false ejects.

Vials
A pharmaceutical manufacturer had roughly 10 million prefilled syringes held in quarantine due to a short-shot defect. Their human inspectors consistently missed the defect, putting product quality, regulatory compliance, and significant revenue at risk.

Syringes
The manufacturer deployed the DAI-50 configured for the automatic inspection of prefilled syringes. Powered by AVIS, the DAI-50 learned to track unpredictable cap motion and reliably detect the nearly impossible to see defect.

Ampoules
With 100% detection accuracy on the short-shot defect, the site implemented the DAI-50 to re-inspect the quarantined product, saving over $3 million in scrap costs.
Next-Gen AI Inspection
Level-Up Your Inspection Process With Our Visual Inspection Machine for Injectable Products
AVIS is the visual inspection platform inside the DAI-50. AVIS uses an unsupervised learning approach that models normal variation rather than relying on defect libraries or labeled images, allowing it to detect both known and unknown anomalies.
Flexible
<60-minutes to train an inspection recipe
Fast
50 parts per minute at maximum speed
Compliant
GMP compliance according to FDA 21 CFR Part 11


Regulatory Requirements for Syringe, Ampoule, and Vial Inspection Machines
The FDA requires 100% of injectable products to undergo visual inspection. Vial, syringe, and ampoule inspection machines must perform as well as, or better than, qualified human inspectors. For years, manufacturers have been stuck: traditional automatic visual inspection equipment is not a viable solution for injectable products with high degrees of normal variability. Consequently, many rely on large teams of human inspectors to meet compliance requirements. AVIS closes this gap by delivering automated visual inspection that performs reliably across complex and difficult-to-inspect products. To date, AVIS has a 100% success rate of outperforming human inspectors in head-to-head testing.
Case Study
98% Detection Accuracy With Our Vial Inspection Machine
AVIS is the visual inspection platform inside the DAI-50. AVIS uses an unsupervised learning approach that models normal variation rather than relying on defect libraries or labeled images, allowing it to detect both known and unknown anomalies.
Challenge
A contract manufacturing organization’s semi-automated vial inspection equipment was failing to detect defects in 20 mL powder-filled molded glass vials due to extreme normal variation in powder disposition and container appearance. This eventually led to AQL failures, production delays, and compliance risk.
Solution
With just 500 compliant units, the DAI-50, powered by AVIS, generated an inspection recipe that captured normal variation in its entirety—delivering automated vial inspection that performs with the flexibility of a human inspector, but with machine-level consistency.
Results
The DAI-50 delivered 98% overall defect-detection accuracy across diverse defect types with a false rejection rate of just ~2.7 %, outperforming traditional methods and supporting improved quality and vial inspection consistency.
Case Study
98% Detection Accuracy With Our Vial Inspection Machine
AVIS is the visual inspection platform inside the DAI-50. AVIS uses an unsupervised learning approach that models normal variation rather than relying on defect libraries or labeled images, allowing it to detect both known and unknown anomalies.
Challenge
A pharmaceutical manufacturer had roughly 10 million prefilled syringes held in quarantine due to a short-shot defect. Their human inspectors consistently missed the defect, putting product quality, regulatory compliance, and significant revenue at risk.
Solution
The manufacturer deployed the DAI-50 configured for the automatic inspection of prefilled syringes. Powered by AVIS, the DAI-50 learned to track unpredictable cap motion and reliably detect the nearly impossible to see defect.
Results
With 100% detection accuracy on the short-shot defect, the site implemented the DAI-50 to re-inspect the quarantined product, saving over $3 million in scrap costs.
Frequently asked questions
Frequently asked questions ordered by popularity. Remember that if the visitor has not committed to the call to action, they may still have questions (doubts) that can be answered.
How does AVIS compare to traditional vial inspection machines?
Compared to traditional high speed inspection machines, AVIS significantly reduces false rejects and lowers the overall false rejection rate across a wide range of difficult-to-inspect pharmaceutical products.
Traditional high speed inspection systems rely on rigid, rule-based programming. To avoid missing defects, they are intentionally conservative — which leads to excessive false rejects. In some cases, traditional systems experience double-digit false rejection rates, especially when inspecting molded glass, powders, lyophilized products, cloudy liquids, or heterogeneous containers.
AVIS, deployed in the DAI-50, uses unsupervised machine learning to model normal variation. Because it understands compliant variability, it reduces unnecessary false rejects across a wide range of container types and product characteristics.
On average, AVIS achieves a false rejection rate as low as 3–4%, even in challenging inspection environments.
How does AVIS help improve patient safety and overall product quality?
Ensuring high quality products at scale requires consistent and reliable quality control. Manual inspection introduces variability due to fatigue, subjectivity, and employee turnover. Traditional automated systems introduce risk through inconsistent defect detection or excessive false rejects.
AVIS strengthens quality control by delivering consistent, automated inspection that performs equal to or better than human inspectors. By reducing falsely accepted defective units and maintaining stable inspection standards, AVIS helps manufacturers consistently produce high quality pharmaceutical products.
What type of machine learning does AVIS use?
AVIS does not rely on deep learning or supervised machine learning models that require thousands of labeled defect images.
Instead, AVIS uses unsupervised machine learning. Rather than training a deep learning classifier on predefined defect categories, AVIS models what “normal” looks like and detects deviations from that baseline.
Most Deep learning approaches depend on large volumes of labeled defect data. In pharmaceutical inspection, defects are rare, inconsistent, and sometimes entirely new. This makes supervised deep learning difficult to scale.
By learning normal variation from approximately 500 compliant units, AVIS can detect both known and previously unseen anomalies, without requiring defect libraries, labeled datasets, or continuous retraining.
